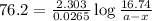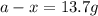Chemistry, 07.03.2020 03:07 canddpotts
The rate constant for this first-order reaction is 0.0265 s − 1 at 300 °C. A ⟶ products If the initial mass of A is 16.74 g, calculate the mass of A remaining after 1.27 min.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Cooldude3966
As you move from right to left on the periodic table the atomic radius fill in the blank
Answers: 2

Chemistry, 22.06.2019 01:00, angelteddy033
Which of the following is not a true statement about dwarf planets? a the kuiper belt contains comets, asteroids, and dwarf planets. b ceres is a dwarf planet located in the kuiper belt. c the largest known dwarf planet in the solar system is named eris.
Answers: 2

Chemistry, 22.06.2019 07:20, camillexv2668
2pos suppose an object in free fall is dropped from a building. its starting velocity is 0 m/s. ignoring the effects of air resistance, what is the speed (in m/s) of the object after falling 3 seconds? give your answer as a positive decimal without units. answer here
Answers: 1

Chemistry, 22.06.2019 10:00, valdezlizbeth6652
Why is carbon ideal for making different compounds?
Answers: 2
You know the right answer?
The rate constant for this first-order reaction is 0.0265 s − 1 at 300 °C. A ⟶ products If the initi...
Questions in other subjects:



Mathematics, 03.03.2021 08:30

Mathematics, 03.03.2021 08:30


History, 03.03.2021 08:30

Mathematics, 03.03.2021 08:30

Mathematics, 03.03.2021 08:30

Mathematics, 03.03.2021 08:30

Social Studies, 03.03.2021 08:30