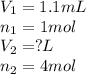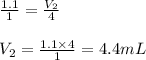Chemistry, 07.03.2020 02:29 homework1911
Methane gas and chlorine gas react to form hydrogen chloride gas and carbon tetrachloride gas. What volume of hydrogen chloride would be produced by this reaction if of methane were consumed? Also, be sure your answer has a unit symbol, and

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, myamiller558
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 09:10, GreatBaconGamer
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1

Chemistry, 22.06.2019 13:50, amandamac7339
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 14:00, rosetoheart2
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2
You know the right answer?
Methane gas and chlorine gas react to form hydrogen chloride gas and carbon tetrachloride gas. What...
Questions in other subjects:

Business, 25.08.2020 01:01



Chemistry, 25.08.2020 01:01


Mathematics, 25.08.2020 01:01


Social Studies, 25.08.2020 01:01

Mathematics, 25.08.2020 01:01

Mathematics, 25.08.2020 01:01

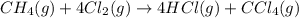
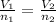
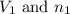 are the volume and number of moles of methane gas
are the volume and number of moles of methane gas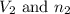 are the volume and number of moles of hydrogen chloride
are the volume and number of moles of hydrogen chloride