
Chemistry, 06.03.2020 02:44 serehnatyras0808
If 1 10 liter sample of O2 gas at 300 kelvin and 0.5 atmosphere of pressure contains 5.0 x 10^22 molecules how many molecules would a 10 liter sample of H2 have if it was at the same temperature and pressure as the oxygen remember that hydrogens are 1/16th the size of oxygen

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:40, Maryjasmine8001
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 14:00, asanchez4292
What type of matter is made of only one kind of atom
Answers: 2

Chemistry, 23.06.2019 01:00, carson9373
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1

Chemistry, 23.06.2019 08:00, vetterk1400
Drag each pressure unit with the corresponding number to describe standard atmospheric pressure
Answers: 1
You know the right answer?
If 1 10 liter sample of O2 gas at 300 kelvin and 0.5 atmosphere of pressure contains 5.0 x 10^22 mol...
Questions in other subjects:


Mathematics, 09.09.2020 20:01








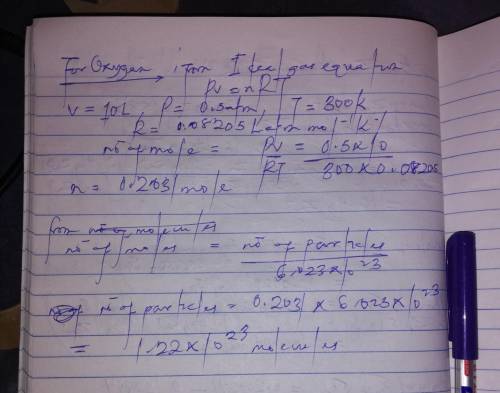
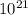
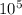 pa
pa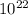 molecules
molecules J/K.
J/K.

