
Chemistry, 06.03.2020 01:48 GhostElite6383
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studying this reaction fills a 7.50 l tank with 29.0 mol of ammonia gas at 35.0 °C. She then raises the temperature, and when the mixture has come to equilibrium measures the amount of nitrogen gas to be 13.0 mol.
Calculate the concentration equilibrium constant for the decomposition of ammonia at the final temperature of the mixture. Round your answer to significant digits.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:30, andrejr0330jr
What is the molar mass of potassium nitrate, kno3
Answers: 1

Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2

Chemistry, 22.06.2019 15:00, makaylajones74pdxtrk
What is the most important factor in determining climates.
Answers: 1

You know the right answer?
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studyin...
Questions in other subjects:


History, 14.12.2019 08:31

Biology, 14.12.2019 08:31


Mathematics, 14.12.2019 08:31


History, 14.12.2019 08:31

Mathematics, 14.12.2019 08:31


History, 14.12.2019 08:31

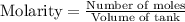

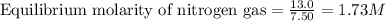
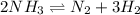

 for above equation follows:
for above equation follows:![K_{eq}=\frac{[N_2][H_2]^3}{[NH_3]^2}](/tpl/images/0535/0025/804f3.png)



