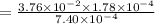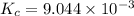Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kiki197701
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 06:30, jonloya264
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 11:30, chelseychew32
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2
You know the right answer?
Some COCl2 is placed in a sealed flask and heated to 756 K. When equilibrium is reached, the flask i...
Questions in other subjects:

English, 21.07.2019 09:30

English, 21.07.2019 09:30


Mathematics, 21.07.2019 09:30

Spanish, 21.07.2019 09:30




Social Studies, 21.07.2019 09:30

Computers and Technology, 21.07.2019 09:30

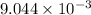 is the value of the equilibrium constant for this reaction at 756 K.
is the value of the equilibrium constant for this reaction at 756 K.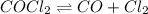
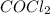
![[COCl_2]=7.40\times 10^{-4} M](/tpl/images/0534/1166/1134d.png)
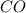
![[CO]=3.76\times 10^{-2} M](/tpl/images/0534/1166/484f5.png)
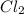
![[Cl_2]=1.78\times 10^{-4} M](/tpl/images/0534/1166/d4643.png)
![K_c=\frac{[CO][Cl_2]}{[COCl_2]}](/tpl/images/0534/1166/59c52.png)