

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, coolkid2041
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1


Chemistry, 22.06.2019 11:10, hannah2757
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1

Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2
You know the right answer?
Carbon tetrachloride can be produced by this reaction: Suppose 1.1 mol and 3.3 mol are placed in a 1...
Questions in other subjects:


Social Studies, 04.07.2019 04:00

History, 04.07.2019 04:00





History, 04.07.2019 04:00


 ⇌
⇌ 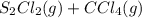
 .
.
 :
:![[CS_2]=\frac{1.1 mol}{1 L}=1.1 M](/tpl/images/0533/8772/cef0e.png)
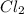 :
:![[Cl_2]=\frac{3.3mol}{1 L}=3.3M](/tpl/images/0533/8772/72054.png)
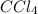 :
:![[CCl_4]=\frac{0.82 mol}{1 L}=0.82 M](/tpl/images/0533/8772/68898.png)
![K_c=\frac{[S_2Cl_2][CCl_4]}{[CS_2][Cl_2]^3}](/tpl/images/0533/8772/40ad0.png)



