Write the balanced net ionic equation for the reactions that occur when the given aqueous solutions are mixed. Include the physical states. A. silver nitrate, AgNO 3 AgNO3 , and magnesium bromide, MgBr 2 MgBr2 net ionic equation: B. perchloric acid, HClO 4 HClO4 , and potassium hydroxide, KOH KOH net ionic equation: C. ammonium sulfide, ( NH 4 ) 2 S (NH4)2S , and cobalt(II) chloride, CoCl 2 CoCl2 net ionic equation:

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 17:30, kiaramccurty
What type of organic molecule comprises the majority of a potato?
Answers: 1


Chemistry, 22.06.2019 21:00, ciel8809
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
You know the right answer?
Write the balanced net ionic equation for the reactions that occur when the given aqueous solutions...
Questions in other subjects:


English, 27.08.2019 04:10

History, 27.08.2019 04:10

Mathematics, 27.08.2019 04:10


Mathematics, 27.08.2019 04:10



Mathematics, 27.08.2019 04:10

History, 27.08.2019 04:10

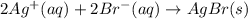
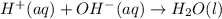


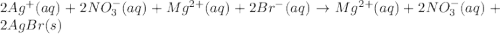
 are the spectator ions.
are the spectator ions.
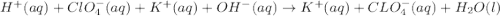
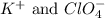 are the spectator ions.
are the spectator ions.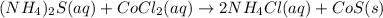
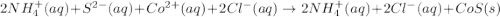
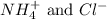 are the spectator ions.
are the spectator ions.