
Chemistry, 03.03.2020 04:00 camiloriveraveoxbgd6
How many moles of sodium acetate must be added to 2.0 L of 0.10 M acetic acid to give a solution that has a pH equal to 5.00? Ignore the volume change due to the addition of sodium acetate.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:00, adjjones2011
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1

Chemistry, 22.06.2019 13:10, bartonamber4042
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
You know the right answer?
How many moles of sodium acetate must be added to 2.0 L of 0.10 M acetic acid to give a solution tha...
Questions in other subjects:





Mathematics, 08.10.2019 06:50


History, 08.10.2019 06:50

Social Studies, 08.10.2019 06:50


![\frac{[acetate]}{[aceticacid]}](/tpl/images/0531/7594/7a095.png)
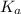 =
= 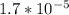
![\frac{[acetate]}{0.1}](/tpl/images/0531/7594/927c3.png)



