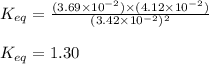Chemistry, 03.03.2020 03:22 icantspeakengles
Some CH2Cl2 is placed in a sealed flask and heated to 517 K. When equilibrium is reached, the flask is found to contain CH2Cl2 (3.42×10-2 M), CH4 (3.69×10-2 M), and CCl4 (4.12×10-2 M). What is the value of the equilibrium constant for this reaction at 517 K?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, micvar9646
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 21.06.2019 23:30, huangjianhe135
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2


Chemistry, 23.06.2019 00:00, maronetham6253
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
Some CH2Cl2 is placed in a sealed flask and heated to 517 K. When equilibrium is reached, the flask...
Questions in other subjects:

Mathematics, 23.04.2020 03:02

Mathematics, 23.04.2020 03:02



Mathematics, 23.04.2020 03:02



English, 23.04.2020 03:02

History, 23.04.2020 03:02

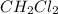 follows:
follows:
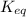 for above equation follows:
for above equation follows:![K_{eq}=\frac{[CH_4][CCl_4]}{[CH_2Cl_2]^2}](/tpl/images/0531/6381/cdd40.png)
![[CH_4]_{eq}=3.69\times 10^{-2}M](/tpl/images/0531/6381/50f3b.png)
![[CCl_4]_{eq}=4.12\times 10^{-2}M](/tpl/images/0531/6381/3e13d.png)
![[CH_2Cl_2]_{eq}=3.42\times 10^{-2}M](/tpl/images/0531/6381/c3f41.png)