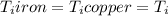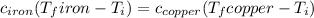Five-gram samples of copper and iron are at room temperature. both samples receive equal amounts of energy due to heat flow. the specific heat capacity of copper is 0.09 cal/g°c, and the specific heat capacity of iron is 0.11 cal/g°c. which of the following statements is true? the temperature of each sample will increase by the same amount. the temperature of each sample will decrease by the same amount. the copper will get hotter than the iron. the iron will get hotter than the copper.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:10, bartonamber4042
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1


Chemistry, 23.06.2019 03:10, 3jazybraxy
Which is true according to the law of conservation of energy
Answers: 1
You know the right answer?
Five-gram samples of copper and iron are at room temperature. both samples receive equal amounts of...
Questions in other subjects:

Mathematics, 09.04.2021 02:50


Mathematics, 09.04.2021 02:50


Spanish, 09.04.2021 02:50


Mathematics, 09.04.2021 02:50

Mathematics, 09.04.2021 02:50

Biology, 09.04.2021 02:50

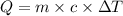
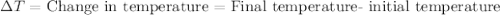
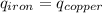
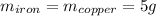 , also
, also