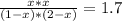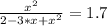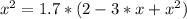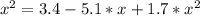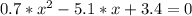Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 08:30, audrey1256
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 11:30, charles8527
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2

You know the right answer?
The reversible chemical reaction A+B⇌C+D has the following equilibrium constant: Kc=[C][D][A][B]=1.7...
Questions in other subjects:


Arts, 17.12.2020 20:30

Mathematics, 17.12.2020 20:30

Mathematics, 17.12.2020 20:30

Mathematics, 17.12.2020 20:30

Mathematics, 17.12.2020 20:30


Physics, 17.12.2020 20:30

History, 17.12.2020 20:30

![Kc=\frac{[C]^{c} *[D]^{d} }{[A]^{a} *[B]^{b} }](/tpl/images/0531/2647/eda24.png)
![Kc=\frac{[C]*[D]}{[A]*[B]}=1.7](/tpl/images/0531/2647/e4842.png)