
Chemistry, 02.03.2020 22:59 hoopstarw4438
At a certain temperature, 3.67 mol SO 2 and 1.83 mol O 2 are placed in a container. 2 SO 2 ( g ) + O 2 ( g ) − ⇀ ↽ − 2 SO 3 ( g ) At equilibrium, there is 1.92 mol SO 3 present. Determine the number of moles of SO 2 and O 2 that are present when the reaction is at equilibrium.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, speris1443
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1


Chemistry, 23.06.2019 01:00, Johnson926
Which elements are found in glucose, the product of photosynthesis? a. carbon, hydrogen, and oxygen b. carbon and hydrogen c. carbon, nitrogen, and oxygen d. hydrogen, nitrogen, and carbon
Answers: 2

Chemistry, 23.06.2019 05:00, cpcoolestkid4
C=59(f−32)the equation above shows how temperature f, measured in degrees fahrenheit, relates to a temperature c, measured in degrees celsius. based on the equation, which of the following must be true? a temperature increase of 1 degree fahrenheit is equivalent to a temperature increase of 59 degree celsius. a temperature increase of 1 degree celsius is equivalent to a temperature increase of 1.8 degrees fahrenheit. a temperature increase of 59 degree fahrenheit is equivalent to a temperature increase of 1 degree celsius. a) i onlyb) ii onlyc) iii onlyd) i and ii only
Answers: 1
You know the right answer?
At a certain temperature, 3.67 mol SO 2 and 1.83 mol O 2 are placed in a container. 2 SO 2 ( g ) + O...
Questions in other subjects:




Social Studies, 02.02.2020 16:04

Business, 02.02.2020 16:04





Chemistry, 02.02.2020 16:04

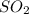 and
and 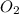 at equilibrium is, 1.75 mol and 0.87 mol respectively.
at equilibrium is, 1.75 mol and 0.87 mol respectively.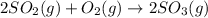
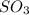 at equilibrium = 1.92
at equilibrium = 1.92

