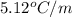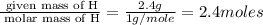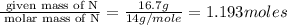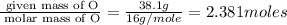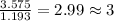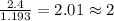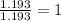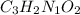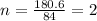Chemistry, 02.03.2020 20:25 EliHarris517
A compound is 42.9% C, 2.4% H, 16.7% N, and 38.1% O, by mass. Addition of 6.45 g of this compound to 50.0 mL benzene, lowers the freezing point from 5.53 to What is the molecular formula of this compound?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, quantamagic
Word equation for k(s)+h2o(l) yield koh(aq) + h2(g)
Answers: 1

Chemistry, 22.06.2019 13:30, makenziehook8
Which is true of a liquid? it has a definite volume but not a definite mass. it has a definite mass but not a definite volume. it has a definite volume but not a definite shape. it has a definite shape but not a definite volume.
Answers: 2

Chemistry, 22.06.2019 16:00, yfnal3x
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
A compound is 42.9% C, 2.4% H, 16.7% N, and 38.1% O, by mass. Addition of 6.45 g of this compound to...
Questions in other subjects:


Mathematics, 05.05.2020 02:34

Mathematics, 05.05.2020 02:34


Social Studies, 05.05.2020 02:34

Mathematics, 05.05.2020 02:34


Biology, 05.05.2020 02:34


Mathematics, 05.05.2020 02:34

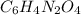
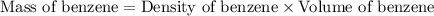
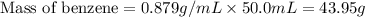
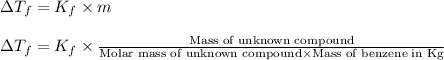
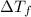 = change in freezing point =
= change in freezing point = 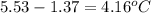
 = freezing point of solution
= freezing point of solution = freezing point of benzene
= freezing point of benzene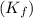 for benzene =
for benzene =