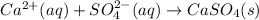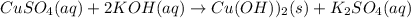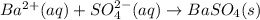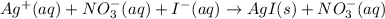Using what you have learned about the three forms of writing chemical equations, classify each of the presented equations as either a molecular equation, a complete ionic equation, or a net ionic equation.
(a) Ca2+(aq) + SO42−(aq)→ CaSO4(s)
(b) CuSO4(aq) + 2KOH(aq)→ Cu(OH)2(s) + K2SO4(aq)
(c) Ba2+ (aq) + SO42−(aq)→ BaSO4(s)
(d) Ag+(aq) + NO3−(aq) + I−(aq)→ AgI(s) + NO3−(aq)

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 01:00, jescanarias22
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 05:30, mandy9386
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 07:20, camillexv2668
2pos suppose an object in free fall is dropped from a building. its starting velocity is 0 m/s. ignoring the effects of air resistance, what is the speed (in m/s) of the object after falling 3 seconds? give your answer as a positive decimal without units. answer here
Answers: 1
You know the right answer?
Using what you have learned about the three forms of writing chemical equations, classify each of th...
Questions in other subjects:








Mathematics, 16.04.2020 21:24

Spanish, 16.04.2020 21:24