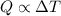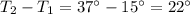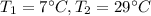Chemistry, 29.02.2020 05:25 kellimcollier8294
A container with a temperature T1 of 15°C is submerged in a bucket of hot water with a temperature T2 of 37°C. An identical container with a temperature of T1 is submerged in an identical bucket of water with a temperature of T2. If the amounts of energy transferred as heat between the containers and the water are the same in both cases, which of the following statements is true?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:30, ashley4329
Select all the correct answers. which compounds have the empirical formula ch20? (multiple answers)a. c2h4o2b. c3h603c. ch2o2d. c5h1005e. c6h1206
Answers: 2

Chemistry, 22.06.2019 23:40, tilievaughn14
What energy conversion occurs when a sling shot is used to shoot a rock across the room? (2 points) question 2 options: 1) stored mechanical energy is converted to mechanical energy. 2) stored mechanical energy is converted to radiant energy. 3) gravitational energy is converted to radiant energy. 4) gravitational energy is converted to mechanical energy.
Answers: 1

Chemistry, 23.06.2019 00:30, quintink
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2
You know the right answer?
A container with a temperature T1 of 15°C is submerged in a bucket of hot water with a temperature T...
Questions in other subjects:



History, 10.11.2020 01:00






Mathematics, 10.11.2020 01:00

Physics, 10.11.2020 01:00