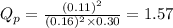Chemistry, 29.02.2020 00:00 KENYONWASHINGTON868
Nitric oxide (NO) reacts readily with chlorine gas as follows.2 NO(g) + Cl2(g) equilibrium reaction arrow 2 NOCl(g)At 700. K the equilibrium constant Kp for this reaction is 0.26. Predict the behavior of each of the following mixtures at this temperature and indicate whether or not the mixtures are at equilibrium. If not, state whether the mixture will need to produce more products or reactants to reach equilibrium.(a) PNO = 0.16 atm, PCl2 = 0.30 atm, and PNOCl = 0.11 atm.(b) PNO = 0.12 atm, PCl2 = 0.10 atm, and PNOCl = 0.048 atm.(c) PNO = 0.15 atm, PCl2 = 0.15 atm, and PNOCl = 5.20 10-3 atm.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:10, glitterpanda2468
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 20:30, camerondillonn
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1

You know the right answer?
Nitric oxide (NO) reacts readily with chlorine gas as follows.2 NO(g) + Cl2(g) equilibrium reaction...
Questions in other subjects:

Mathematics, 28.09.2020 19:01


Mathematics, 28.09.2020 19:01


Mathematics, 28.09.2020 19:01

Chemistry, 28.09.2020 19:01

Mathematics, 28.09.2020 19:01


Mathematics, 28.09.2020 19:01

History, 28.09.2020 19:01


 for above equation follows:
for above equation follows: .....(1)
.....(1) = 0.26
= 0.26 ; the reaction is product favored.When
; the reaction is product favored.When 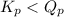 ; the reaction is reactant favored.When
; the reaction is reactant favored.When 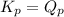 ; the reaction is in equilibrium.
; the reaction is in equilibrium.