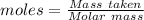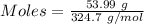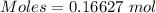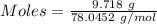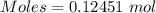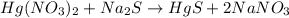Chemistry, 28.02.2020 23:26 anasstasiyashpa
If a solution containing 53.99 g 53.99 g of mercury(II) nitrate is allowed to react completely with a solution containing 9.718 g 9.718 g of sodium sulfide, how many grams of solid precipitate will form?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, danielhall
Identify a strong intermolecular force of attraction between an alcohol
Answers: 1

Chemistry, 22.06.2019 19:00, georgesarkes12
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2


Chemistry, 23.06.2019 06:10, ridzrana02
How can liquids be seperated by density a the liquids are absorbed onto a paper b the liquids are turned into seperate vapors c the liquids are collected as they evaporate d the liquids are allowed to seperate into layers
Answers: 1
You know the right answer?
If a solution containing 53.99 g 53.99 g of mercury(II) nitrate is allowed to react completely with...
Questions in other subjects:





Biology, 22.04.2020 02:27

Computers and Technology, 22.04.2020 02:27