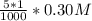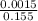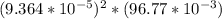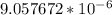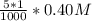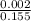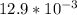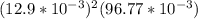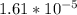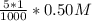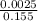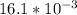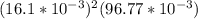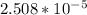For which of the following mixtures will Ag2SO4(s) precipitate?
a. 150.0 mL of 0.10 M Na2SO4(aq) and 5.0 mL of 0.20 M AgNO3(aq)
b. 150.0 mL of 0.10 M Na2SO4(aq) and 5.0 mL of 0.30 M AgNO3(aq)
c. 150.0 mL of 0.10 M Na2SO4(aq) and 5.0 mL of 0.40 M AgNO3(aq)
d. 150.0 mL of 0.10 M Na2SO4(aq) and 5.0 mL of 0.50 M AgNO3(aq)

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 01:30, rubyr9975
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1
You know the right answer?
For which of the following mixtures will Ag2SO4(s) precipitate?
a. 150.0 mL of 0.10 M Na2SO4(a...
a. 150.0 mL of 0.10 M Na2SO4(a...
Questions in other subjects:

Mathematics, 16.03.2020 20:53

Social Studies, 16.03.2020 20:53





Health, 16.03.2020 20:53

Biology, 16.03.2020 20:53


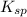 =
= ![[Ag^+]^2[SO_4^{2-}]](/tpl/images/0527/4149/191c0.png)
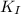 <
< 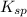 ; precipitation will not occur
; precipitation will not occur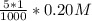
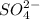 =
= 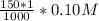
![[Ag^+]^2](/tpl/images/0527/4149/1aebd.png) =
= 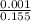

![[SO_4^{2-}]](/tpl/images/0527/4149/43b69.png) =
= 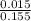

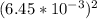 ×
×