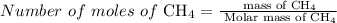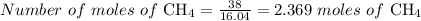2.3 moles of 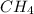
Option B
Explanation:
Mole is the standard unit used to express the amount of a chemical compounds. To find the number of moles of any chemical compound, divide the mass by the molar mass of that chemical compound. The equation is given by,
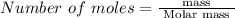
Given data:
Mass of methane = 38 grams
we know, the molar mass of methane = 16.04 g/mole