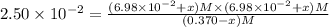Chemistry, 27.02.2020 20:13 SKYBLUE1015
Some SbCl5 is allowed to dissociate into SbCl3 and Cl2 at 521 K. At equilibrium, [SbCl5] = 0.195 M, and [SbCl3] = [Cl2] = 6.98×10-2 M. Additional SbCl5 is added so that [SbCl5]new = 0.370 M and the system is allowed to once again reach equilibrium.
SbCl5(g) <-> SbCl3(g) + Cl2(g) K = 2.50×10-2 at 521 K
(a) In which direction will the reaction proceed to reach equilibrium? to the right to the left
(b) What are the new concentrations of reactants and products after the system reaches equilibrium?
[SbCl5] = M
[SbCl3] = ___M
[Cl2] = ___M

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 13:00, torigirl4126
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3


Chemistry, 22.06.2019 22:30, jkjjoijjm5928
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3
You know the right answer?
Some SbCl5 is allowed to dissociate into SbCl3 and Cl2 at 521 K. At equilibrium, [SbCl5] = 0.195 M,...
Questions in other subjects:

Mathematics, 01.04.2020 16:59





Business, 01.04.2020 17:00




![[SbCl_5]=(0.370-x) M=(0.370-0.0233) M=0.3467 M](/tpl/images/0527/0501/9b64f.png)
![[SbCl_3]=(6.98\times 10^{-2}+x) M=(6.98\times 10^{-2}+0.0233) M=0.0931 M](/tpl/images/0527/0501/4f238.png)
![[Cl_2]=(6.98\times 10^{-2}+x) M=(6.98\times 10^{-2}+0.0233) M=0.0931 M](/tpl/images/0527/0501/5868d.png)
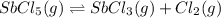
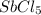 is increasing .So, the equilibrium will shift in the right direction.
is increasing .So, the equilibrium will shift in the right direction.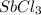 =
= 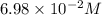
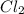 =
= 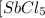 to 0.370 M at equilibrium :
to 0.370 M at equilibrium :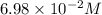
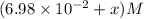
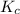
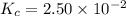
![K_c=\frac{[SbCl_3][Cl_2]}{[SbCl_5]}](/tpl/images/0527/0501/c5c78.png)