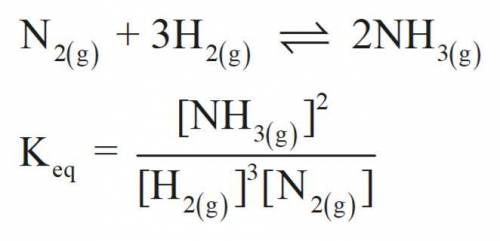Chemistry, 27.02.2020 19:26 Batzs3rdacct
Consider the following chemical equilibrium:
N2(g) + 3H2(g) ⇌ 2NH3
Now write an equation below that shows how to calculate from for this reaction at an absolute temperature . You can assume is comfortably above room temperature.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, kathleensumter4913
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 10:10, jojomgarcia01
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2
You know the right answer?
Consider the following chemical equilibrium:
N2(g) + 3H2(g) ⇌ 2NH3
Now writ...
N2(g) + 3H2(g) ⇌ 2NH3
Now writ...
Questions in other subjects:







Computers and Technology, 11.01.2020 02:31