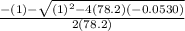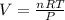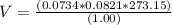Consider the reaction below for which K = 78.2 atm-1. A(g) + B(g) ↔ C(g) Assume that 0.386 mol C(g) is placed in the cylinder represented below. The barometric pressure on the piston (which is assumed to be massless and frictionless) is constant at 1.00 atm. The original volume (before the 0.386 mol C(g) begins to decompose) is 7.29 L. The temperature is fixed and can be determined from the idea gas law. What is the volume in the cylinder at equilibrium?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, mgavyn1052
Write a paragraph that provides examples of each stage of volcanic activity, a description of the volcano, and facts about each stage.
Answers: 1

Chemistry, 22.06.2019 04:30, coryoddoc3685
Turbo the snail moves across the ground at a pace of 12 feet per day. if the garden is 48 feet away, how many days will it take for the snail to get there?
Answers: 2

You know the right answer?
Consider the reaction below for which K = 78.2 atm-1. A(g) + B(g) ↔ C(g) Assume that 0.386 mol C(g)...
Questions in other subjects:


English, 29.10.2019 18:31

Social Studies, 29.10.2019 18:31

Mathematics, 29.10.2019 18:31







![K = \frac{[C]}{[A][B]}](/tpl/images/0526/7975/63063.png)
![78.2=\frac{[0.0530-x]}{[x][x]}](/tpl/images/0526/7975/8334d.png)
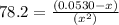
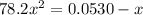
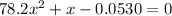
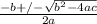
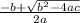 or
or 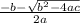
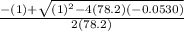 or
or