
Calcium nitrate will react with ammonium chloride at slightly elevated temperatures, as represented in the equation below. Ca(NO3)2(s) + 2NH4Cl(s) → 2N2O(g) + CaCl2(s) + 4H2O(g) What is the maximum volume of N2O at STP that could be produced using a 5.20-mol sample of each reactant?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, Homepage10
Theoretically, which metal should be the most reactive? hydrogen lithium francium fluorine
Answers: 1



Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2
You know the right answer?
Calcium nitrate will react with ammonium chloride at slightly elevated temperatures, as represented...
Questions in other subjects:



Arts, 02.04.2020 03:26

Mathematics, 02.04.2020 03:26






Mathematics, 02.04.2020 03:26

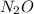 at STP produced is, 116.48 L
at STP produced is, 116.48 L
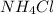 react with 1 mole of
react with 1 mole of 
 moles of
moles of 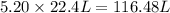 volume of gas
volume of gas 

