
Chemistry, 27.02.2020 00:13 meganwintergirl
Suppose 5.00 moles of Lithium Nitrate react with 5.00 moles of Magnesium Fluoride react according to **BALANCE 1st!!**: __Li(NO3) __ MgF2 -> __LiF __Mg(NO3)2

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, ebigham5117
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

Chemistry, 22.06.2019 20:30, huangjianhe135
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 23.06.2019 06:00, wirchakethan23
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
Suppose 5.00 moles of Lithium Nitrate react with 5.00 moles of Magnesium Fluoride react according to...
Questions in other subjects:

Mathematics, 17.12.2020 01:40




History, 17.12.2020 01:40



Mathematics, 17.12.2020 01:40

Computers and Technology, 17.12.2020 01:40

Mathematics, 17.12.2020 01:40

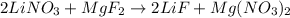
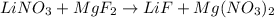
 and
and  then we get the balanced chemical equation.
then we get the balanced chemical equation.

