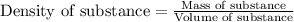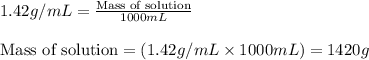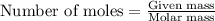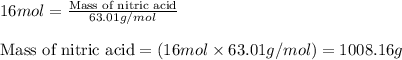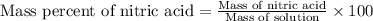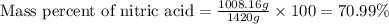Chemistry, 26.02.2020 23:49 garrettrhoad
Ommercially available aqueous nitric acid has a density of 1.42 g/mL and a concentration of 16 M. Calculate the mass percent of HNO3 (molar mass = 63.01 g/mol) in the solution.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, SmolBeanPotato
What is the volume of the fluid in the graduated cylinder with accuracy and measured to the correct degree of precision? 41.2 ml 42.0 ml 41.23 ml 41.89 ml
Answers: 1

Chemistry, 22.06.2019 05:00, Angelanova69134
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Chemistry, 22.06.2019 09:30, junkmailemail42
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 12:20, jessicasbss6840
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1
You know the right answer?
Ommercially available aqueous nitric acid has a density of 1.42 g/mL and a concentration of 16 M. Ca...
Questions in other subjects:

Mathematics, 29.03.2020 01:35


Mathematics, 29.03.2020 01:35


Biology, 29.03.2020 01:35



Mathematics, 29.03.2020 01:35

Mathematics, 29.03.2020 01:35

Mathematics, 29.03.2020 01:35