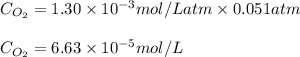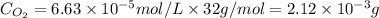Chemistry, 26.02.2020 23:27 josephrosanne18
The partial pressure of in your lungs varies from 25 mm Hg to 40 mm Hg. What mass of can dissolve in 1.0 L of water at 25 °C if the partial pressure of is 39 mm Hg?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, brittanysanders
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2


Chemistry, 22.06.2019 11:00, blondieb1722
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2

Chemistry, 22.06.2019 17:00, smelcher3900
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2
You know the right answer?
The partial pressure of in your lungs varies from 25 mm Hg to 40 mm Hg. What mass of can dissolve in...
Questions in other subjects:


History, 12.12.2020 16:00

Social Studies, 12.12.2020 16:00

Biology, 12.12.2020 16:00

Mathematics, 12.12.2020 16:00


Mathematics, 12.12.2020 16:00

English, 12.12.2020 16:00

English, 12.12.2020 16:00

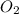 in your lungs varies from 25 mm Hg to 40 mm Hg. What mass of
in your lungs varies from 25 mm Hg to 40 mm Hg. What mass of 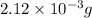
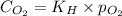
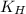 = Henry's constant =
= Henry's constant = 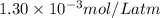
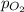 = partial pressure = 39 mm Hg = 0.051 atm (760mmHg=1atm)
= partial pressure = 39 mm Hg = 0.051 atm (760mmHg=1atm)