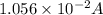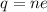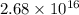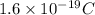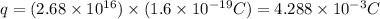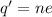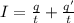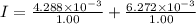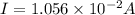Chemistry, 26.02.2020 19:12 karenjunior
Current passes through a solution of sodium chloride. In 1.00 second, 2.68×1016Na+ ions arrive at the negative electrode and 3.92×1016Cl− ions arrive at the positive electrode. Part A What is the current passing between the electrodes?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:50, iloveballet1857
The electron configuration for chromium is 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 3 d 5 4 s 1 instead of 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 3 d 4 4 s 1 . the configuration is an exception to the
Answers: 3

Chemistry, 22.06.2019 00:00, brookemcelhaney
Which of the following methods uses the decay of atomic particles in an object to find its exact age? a. fossil dating b. geologic dating c. radioactive dating d. relative dating
Answers: 1

Chemistry, 22.06.2019 10:50, mi364
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3
You know the right answer?
Current passes through a solution of sodium chloride. In 1.00 second, 2.68×1016Na+ ions arrive at th...
Questions in other subjects:


Biology, 02.08.2019 15:30

Geography, 02.08.2019 15:30


Biology, 02.08.2019 15:30

History, 02.08.2019 15:30

Geography, 02.08.2019 15:30

English, 02.08.2019 15:30

Geography, 02.08.2019 15:30