
Chemistry, 26.02.2020 01:54 xxtonixwilsonxx
Consider the following system at equilibrium:A(aq)+B(aq) <---> 2C(aq)Classify each of the following actions by whether it causes a leftward shift, a rightward shift, or no shift in the direction of the net reaction.1. Increase A- Right2. Increase B- Right3. Increase C- Left4. Decrease A- Right5. Decrease B- Right6. Decrease C- lEft7. Double A and Halve B- NO Shift8. Double both B and C- NO shift

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:50, ayoismeisjjjjuan
Suppose you got a low yield of benzoin from your benzoin condensation reaction and thus only have 0.300 g of benzoin to use as the starting material for this reaction. how much concentrated nitric acid should you add? (concentrated nitric acid is 15.8 m). write your answer in the form x. xx ml
Answers: 1


Chemistry, 22.06.2019 10:00, emfranco1
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 12:10, kaitlynbernatz2778
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3
You know the right answer?
Consider the following system at equilibrium:A(aq)+B(aq) <---> 2C(aq)Classify each of the foll...
Questions in other subjects:


English, 26.01.2020 05:31

Mathematics, 26.01.2020 05:31

French, 26.01.2020 05:31


Biology, 26.01.2020 05:31

Mathematics, 26.01.2020 05:31


Social Studies, 26.01.2020 05:31

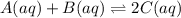
![K=\frac{[C]^2}{[A][B]}](/tpl/images/0524/2481/477c9.png)
![K'=\frac{[C]^2}{[2A][\frac{B}{2}]}=\frac{[C]^2}{[A][B]}](/tpl/images/0524/2481/7596a.png)
![K'=\frac{[2C]^2}{[A][2B]}=\frac{4[C]^2}{[A][2B]}=\frac{2[C]^2}{[A][B]}](/tpl/images/0524/2481/c241a.png)


