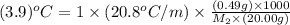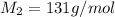Chemistry, 21.02.2020 22:44 princessammarah4731
When 0.49 g of a molecular compound was dissolved in 20.00 g of cyclohexane, the freezing point of the solution was lowered by 3.9 0C. Determine the molecular mass of this compound.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, lukeakalucas
Alarge marble is dropped in a graduated cylinder with 35ml of water in it. the water level increases to 49ml. what is the volume of the marble
Answers: 1

Chemistry, 22.06.2019 19:00, innocentman69
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 23.06.2019 01:30, nikonee
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1
You know the right answer?
When 0.49 g of a molecular compound was dissolved in 20.00 g of cyclohexane, the freezing point of t...
Questions in other subjects:

Mathematics, 19.11.2020 23:30





Biology, 19.11.2020 23:30




Mathematics, 19.11.2020 23:30

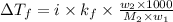
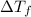 = depression in freezing point =
= depression in freezing point = 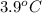
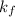 = freezing point constant =
= freezing point constant = 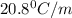
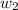 = mass of solute = 0.49 g
= mass of solute = 0.49 g
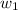 = mass of solvent (cyclohexane) = 20.00 g
= mass of solvent (cyclohexane) = 20.00 g
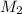 = molar mass of solute = ?
= molar mass of solute = ?