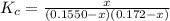Chemistry, 21.02.2020 21:58 leahstubbs
For the following reaction, Kc = 255 at 1000 K. CO(g)+Cl2(g)⇌COCl2(g) A reaction mixture initially contains a CO concentration of 0.1550 M and a Cl2 concentration of 0.172 M at 1000 K.
What is the equilibrium concentration of CO at 1000 K?
What is the equilibrium concentration of Cl2 at 1000 K?
What is the equilibrium concentration of COCl2 at 1000 K?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, ElizabethF
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3


You know the right answer?
For the following reaction, Kc = 255 at 1000 K. CO(g)+Cl2(g)⇌COCl2(g) A reaction mixture initially c...
Questions in other subjects:



History, 24.06.2019 19:00

History, 24.06.2019 19:00

Social Studies, 24.06.2019 19:00

History, 24.06.2019 19:00

Mathematics, 24.06.2019 19:00


Mathematics, 24.06.2019 19:00


 initial
initial![K_c = \frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0519/6554/f9140.png)
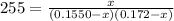
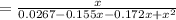
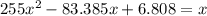
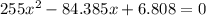

 (correct value)
(correct value)

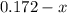


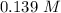
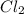 at 1000 K is= 0.033 M and the equilibrium concentration of
at 1000 K is= 0.033 M and the equilibrium concentration of  at 1000 K is 0.139 M
at 1000 K is 0.139 M 
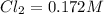

![K_c=\frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0519/6554/36d91.png)