
Chemistry, 21.02.2020 17:42 macylen3900
Air bags are activated when a severe impact causes a steel ball to compress a spring and electrically ignite a detonator cap. This action causes sodium azide (NaN3) to decompose explosively according to the following reaction. 2 NaN3(s) → 2 Na(s) + 3 N2(g) What mass of NaN3(s) must be reacted to inflate an air bag to 70.6 L at STP?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, ayoismeisalex
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1



Chemistry, 23.06.2019 02:00, Hellopeople233
Which of the following substances is the most soluble in water? a. sodium chloride b. methane c. bromine d. carbon
Answers: 1
You know the right answer?
Air bags are activated when a severe impact causes a steel ball to compress a spring and electricall...
Questions in other subjects:

History, 10.12.2020 19:30

Mathematics, 10.12.2020 19:30

Chemistry, 10.12.2020 19:30

Chemistry, 10.12.2020 19:30

Mathematics, 10.12.2020 19:30


Chemistry, 10.12.2020 19:30



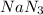 must be reacted to inflate an air bag to 70.6 L at STP.
must be reacted to inflate an air bag to 70.6 L at STP.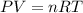
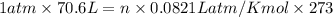
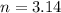
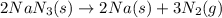
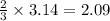 moles of
moles of 

