
Please use the values in the resources listed below instead of the textbook values. Under certain conditions the decomposition of ammonia on a metal surface gives the following data.[NH3] (M) 2.0 ✕ 10−3 4.0 ✕ 10−3 6.0 ✕ 10−3 Rate (mol/L/h) 1.5 ✕ 10−6 1.5 ✕ 10−6 1.5 ✕ 10−6 Determine the rate equation for this reaction. (Rate expressions take the general form: rate = k . [A]a . [B]b.)

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, kingbot350
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

You know the right answer?
Please use the values in the resources listed below instead of the textbook values. Under certain co...
Questions in other subjects:

History, 21.09.2020 05:01









![R=k[NH_3]^0](/tpl/images/0518/3680/fda84.png)
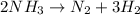
![R=k[NH_3]^x](/tpl/images/0518/3680/194a4.png)
![[NH_3]=2.0\times 10^{-3} M](/tpl/images/0518/3680/fed44.png)
![1.5\times 10^{-6} M/s=k[2.0\times 10^{-3} M]^x](/tpl/images/0518/3680/2ac63.png) ..[1]
..[1]![[NH_3]=4.0\times 10^{-3} M](/tpl/images/0518/3680/8a2bc.png)
![1.5\times 10^{-6} M/s=k[4.0\times 10^{-3} M]^x](/tpl/images/0518/3680/49a41.png) ..[2]
..[2]![\frac{1.5\times 10^{-6}M/s}{1.5\times 10^{-6}M/s}=\frac{k[2.0\times 10^{-3}M]^x}{k[4.0\times 10^{-3}M]^x}](/tpl/images/0518/3680/c6074.png)


