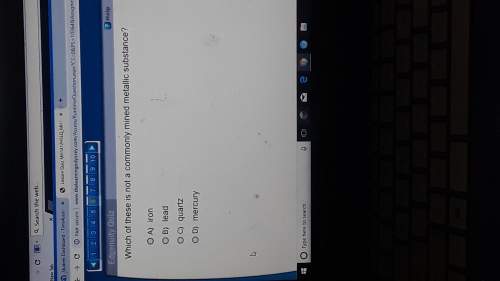
Chemistry, 20.02.2020 08:02 cjhenson02
A 1.0108 g sample of an unknown nonelectrolyte is dissolved in 10.0090 g of benzophenone and produces a solution that freezes at 35.8 C. If the pure benzophenone melted at 48.1 C, what is the molecular weight of the unknown compound?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, nuclearfire278
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease. correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 10:00, zionlopez543
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1
You know the right answer?
A 1.0108 g sample of an unknown nonelectrolyte is dissolved in 10.0090 g of benzophenone and produce...
Questions in other subjects:

Mathematics, 19.07.2019 18:00


Mathematics, 19.07.2019 18:00

Mathematics, 19.07.2019 18:00

Business, 19.07.2019 18:00



Business, 19.07.2019 18:00


Health, 19.07.2019 18:00