
Chemistry, 20.02.2020 01:10 gabrielolivas59
The percentage deprotonation of a 0.115 M solution of benzoic acid (a weak, monoprotic acid) is 2.1%. What is the pH of the solution

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, anglacx5465
Why are pipes bursting in the in extremely cold weather?
Answers: 2

Chemistry, 22.06.2019 22:30, angelagonzalesownus1
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1

Chemistry, 23.06.2019 00:40, joe7977
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
The percentage deprotonation of a 0.115 M solution of benzoic acid (a weak, monoprotic acid) is 2.1%...
Questions in other subjects:



Geography, 25.07.2019 02:10







English, 25.07.2019 02:10

 =
= 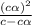 ( HA ---> H⁺A⁻ )
( HA ---> H⁺A⁻ )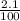 = 0.021
= 0.021 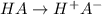
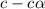
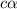
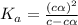
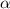 =
= 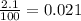
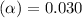
![[H^+]=c\times \alpha](/tpl/images/0516/4496/4fc41.png)
![[H^+]=0.115\times 0.021=0.0024](/tpl/images/0516/4496/7e639.png)
![pH=-log[H^+]](/tpl/images/0516/4496/15713.png)
![pH=-log[0.0024]=2.6](/tpl/images/0516/4496/aa86c.png)


