
Chemistry, 18.02.2020 21:22 leandrogarin37p2g5ds
Phosphoric acid is a triprotic acid ( K a1 = 6.9 × 10 − 3 Ka1=6.9×10−3, K a2 = 6.2 × 10 − 8 Ka2=6.2×10−8, and K a3 = 4.8 × 10 − 13 Ka3=4.8×10−13). To find the pH of a buffer composed of H 2 PO − 4 ( aq ) H2PO4−(aq) and HPO 2 − 4 ( aq ) HPO42−(aq) , which p K a Ka value should be used in the Henderson–Hasselbalch equation?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 21:50, isabel81ie
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l. s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2

You know the right answer?
Phosphoric acid is a triprotic acid ( K a1 = 6.9 × 10 − 3 Ka1=6.9×10−3, K a2 = 6.2 × 10 − 8 Ka2=6.2×...
Questions in other subjects:


History, 26.03.2021 20:10


Mathematics, 26.03.2021 20:10




Biology, 26.03.2021 20:10

Biology, 26.03.2021 20:10

Mathematics, 26.03.2021 20:10

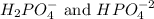 , we use the
, we use the 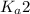
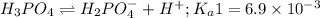
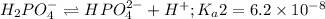
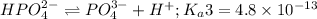
 of second dissociation process
of second dissociation process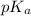 , we use the equation:
, we use the equation: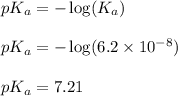
![pH=pK_a2+\log(\frac{[\text{conjugate base}]}{[\text{weak acid}]})](/tpl/images/0514/6294/e7d91.png)
![pH=pK_a2+\log(\frac{[HPO_4^{2-}]}{[H_2PO_4^-]})](/tpl/images/0514/6294/f63a6.png)
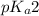 = negative logarithm of second acid dissociation constant of phosphoric acid = 7.21
= negative logarithm of second acid dissociation constant of phosphoric acid = 7.21![[HPO_4^{2-}]](/tpl/images/0514/6294/c0ca9.png) = concentration of conjugate base
= concentration of conjugate base![[H_2PO_4^{-}]](/tpl/images/0514/6294/fcc52.png) = concentration of weak acid
= concentration of weak acid

