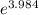The activation energy for the isomerization ol cyclopropane to propene is 274 kJ/mol. By what factor does the rate of this reaction increase as the temperature rises from 255 to 291 oC? Hint: The factor is the ratio of the rates. Since rate is directly proportional to rate constant, the factor is also ratio of rate constants.

Answers: 3


Other questions on the subject: Chemistry




Chemistry, 23.06.2019 08:30, alexiasommers41
Imagine you are a business executive who wants to pursue an environment policy for your company that limits pollution and uses fewer raw materials but would cost more what might be the discussion to your next broad meeting how would you make your case to your shareholders
Answers: 1
You know the right answer?
The activation energy for the isomerization ol cyclopropane to propene is 274 kJ/mol. By what factor...
Questions in other subjects:

Chemistry, 01.09.2019 06:20


History, 01.09.2019 06:20


Mathematics, 01.09.2019 06:20



Mathematics, 01.09.2019 06:20

Health, 01.09.2019 06:20

Physics, 01.09.2019 06:20

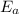 ) = 274 kJ/mol.
) = 274 kJ/mol. 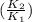 =
= ![\frac{E_a}{R} [\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0514/4438/3b4c3.png)
![\frac{273kJ/mol}{8.314*10^{-3}kJK^{-1}} [\frac{1}{528}-\frac{1}{564}]](/tpl/images/0514/4438/e9d63.png)
![\frac{273kJ/mol}{8.314*10^{-3}kJK^{-1}} [\frac{36k}{(528K)(564K)}]](/tpl/images/0514/4438/2ab49.png)