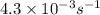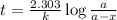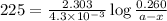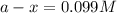Chemistry, 18.02.2020 19:16 dajmoneyp80a0o
The following reaction was monitored as a function of time: A→B+C A plot of ln[A] versus time yields a straight line with slope −4.3×10−3 /s. If the initial concentration of A is 0.260 M, what is the concentration after 225 s?

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, MansellS5529
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 22.06.2019 05:40, timmonskids6027
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1
You know the right answer?
The following reaction was monitored as a function of time: A→B+C A plot of ln[A] versus time yields...
Questions in other subjects:



Mathematics, 06.03.2020 15:42

History, 06.03.2020 15:43




Computers and Technology, 06.03.2020 15:43