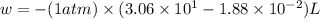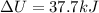Chemistry, 18.02.2020 02:18 emily12403
At 373.15K and 1 atm, the molar volume of liquid water and steamare 1.88 X 10-5 m3 and 3.06 X 10-2m3, respectively. Given that the heat of vaporization ofwater is 40.79 kJ/mol, calculate the values of ?H and ?Ufor 1 mole in the following process:
H2O (l, 373.15 K, 1 atm) ---> H2O(g, 373.15 K, 1 atm)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:40, justicejesusfreak
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1


Chemistry, 22.06.2019 15:30, abdullaketbi71
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1

Chemistry, 22.06.2019 18:50, cj31150631
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1
You know the right answer?
At 373.15K and 1 atm, the molar volume of liquid water and steamare 1.88 X 10-5 m3 and 3.06 X 10-2m3...
Questions in other subjects:



Mathematics, 19.03.2021 01:50


Mathematics, 19.03.2021 01:50




Mathematics, 19.03.2021 01:50

Mathematics, 19.03.2021 01:50

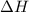 and
and 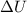 is, 40.79 kJ and 37.7 kJ respectively.
is, 40.79 kJ and 37.7 kJ respectively.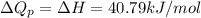

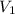 = initial volume =
= initial volume = 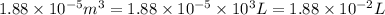
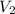 = final volume =
= final volume =