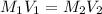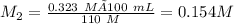Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, chloe081
We just started a new lesson in chemistry and everyone hates it and i dont get it one bit. i hate school. h el p. balanced equationc3h8+5o2-> 3co2+4h2o1.) if you start with 14.8g of propane(c3h8) and 3.44g of oxygen, which is the limiting reactant -check my answer 2.)what mass of excess reagent is left over? 3.)what mass of carbon dioxide can be made? 4.)what mass of water is produced?
Answers: 2

Chemistry, 22.06.2019 02:00, rosie20052019
Which of the following happens during cell division? (a) energy is created (b) waste is eliminated (c) carbon dioxide is released (d) damaged cells are replaced
Answers: 1


Chemistry, 23.06.2019 06:20, ratpizza
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1
You know the right answer?
A 100. mL solution of NaOH has a pH of 13.51. If you add 110 mL of water to the original solution, w...
Questions in other subjects:







English, 24.03.2020 00:32

Mathematics, 24.03.2020 00:32


![[OH]=10^{-pOH}\\=10^{0.49}= 0.323 M](/tpl/images/0513/6468/22218.png)