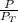A mixture of ethanol and 1−propanol behaves ideally at 36°C and is in equilibrium with its vapor. If the mole fraction of ethanol in the solution is 0.65, calculate its mole fraction in the vapor phase at this temperature. (The vapor pressures of pure ethanol and 1−propanol at 36°C are 108 and 40.0 mmHg, respectively.)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3


Chemistry, 23.06.2019 05:10, citlalli30
Name a brittle metal , which is used to galvanize iron
Answers: 1

Chemistry, 23.06.2019 12:30, jamarstand
D5w is shorthand for a 5% glucose rehydration fluid used in ivs. the doctor orders 1200 ml d5w@ 30 gtts/min. you have an iv tube that delivers 18 gtts/cc. how many hours will it take for the 1200 cc bottle to infuse?
Answers: 2
You know the right answer?
A mixture of ethanol and 1−propanol behaves ideally at 36°C and is in equilibrium with its vapor. If...
Questions in other subjects:


History, 01.01.2020 08:31

History, 01.01.2020 08:31

Mathematics, 01.01.2020 08:31

English, 01.01.2020 08:31


Chemistry, 01.01.2020 08:31

Physics, 01.01.2020 08:31


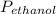 = 0.65 × 108 mmHg
= 0.65 × 108 mmHg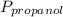 = 0.35× 40.0 mmHg
= 0.35× 40.0 mmHg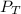 = 70.2 + 4.9 = 75.1 mmHg
= 70.2 + 4.9 = 75.1 mmHg