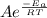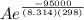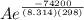The standard free energy of activation of one reaction A is 95.00 kJ mol–1 (22.71 kcal mol–1). The standard free energy of activation of another reaction B is 74.20 kJ mol–1 (17.73 kcal mol–1). Assume a temperature of 298 K and 1 M concentration.
By what factor is one reaction faster than the other?
Which reaction is faster?
(a) Reaction A is faster.
(b) Reaction B is faster.
(c) Cannot be determined.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:00, devontemiles8868
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 22.06.2019 13:00, nauticatyson9
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
You know the right answer?
The standard free energy of activation of one reaction A is 95.00 kJ mol–1 (22.71 kcal mol–1). The s...
Questions in other subjects:


Mathematics, 02.03.2020 05:32

Social Studies, 02.03.2020 05:32

Mathematics, 02.03.2020 05:32

Physics, 02.03.2020 05:33





Chemistry, 02.03.2020 05:35