
A 3.76 g sample of a compound consisting of carbon, hydrogen, oxygen, nitrogen, and sulfur was combusted in excess oxygen. This produced 4.06 g CO 2 and 2.22 g H 2 O . A second sample of this compound with a mass of 5.69 g produced 3.73 g SO 3 . A third sample of this compound with a mass of 8.53 g produced 4.40 g HNO 3 . Determine the empirical formula of the compound. Enter the correct subscripts on the given chemical formula.?

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 21:00, taylorlanehart
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
You know the right answer?
A 3.76 g sample of a compound consisting of carbon, hydrogen, oxygen, nitrogen, and sulfur was combu...
Questions in other subjects:



Advanced Placement (AP), 18.11.2020 01:00


Mathematics, 18.11.2020 01:00




Mathematics, 18.11.2020 01:00

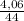 = 0,092g/mol
= 0,092g/mol . 100 =
. 100 = 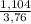 . 100 = 29,4%
. 100 = 29,4%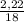 = 0,124 g/mol
= 0,124 g/mol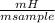 . 100 =
. 100 =  . 100 = 6,6%
. 100 = 6,6%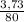 = 0,046g/mol
= 0,046g/mol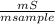 . 100 =
. 100 = 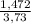 . 100 = 39,5%
. 100 = 39,5%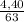 = 0,07
= 0,07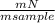 . 100 =
. 100 = 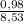 . 100 = 11,5%
. 100 = 11,5%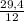 = 2,45 mol
= 2,45 mol

