1. ClO-(aq) + H2O(l) => HClO(aq) + OH-(aq) SLOW

Chemistry, 13.02.2020 18:36 samantha9430
The proposed mechanism for a reaction is
1. ClO-(aq) + H2O(l) => HClO(aq) + OH-(aq) SLOW
2. I-(aq) + HClO(aq) => HIO(aq) + Cl-(aq) FAST
3. OH-(aq) + HIO(aq) => H2O(l) + IO-(aq) FAST
Which of the following would be a rate law for the reaction?
1. rate = k[ClO-][H2O][I-][OH-]
2. rate = k[ClO-][H2O]
3. rate = k[OH-][HIO]
4. rate = k[I-][HClO]
5. rate = k[ClO-][H2O][I-]

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, caeyanij
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3

Chemistry, 22.06.2019 09:30, mimibear2932
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

You know the right answer?
The proposed mechanism for a reaction is
1. ClO-(aq) + H2O(l) => HClO(aq) + OH-(aq) SLOW
1. ClO-(aq) + H2O(l) => HClO(aq) + OH-(aq) SLOW
Questions in other subjects:


Mathematics, 10.03.2020 07:50

Mathematics, 10.03.2020 07:50

Social Studies, 10.03.2020 07:50






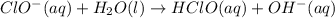 is a slow step reaction.
is a slow step reaction.![k[ClO^{-}][H_{2}O]](/tpl/images/0510/0437/87324.png)


