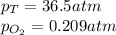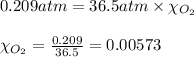Chemistry, 13.02.2020 05:14 jrassicworld4ever
What should be the mole fraction of O2 in the gas mixture the diver breathes in order to have the same partial pressure of oxygen in his lungs as he would at sea level? Note that the mole fraction of oxygen at sea level is 0.209.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:40, ricardoamora54
Who is better, messi or cristiano, i need this for a chemistry class. asap
Answers: 1

Chemistry, 22.06.2019 00:10, scottbrandon653
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2


Chemistry, 22.06.2019 14:50, ladybugperez05
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1
You know the right answer?
What should be the mole fraction of O2 in the gas mixture the diver breathes in order to have the sa...
Questions in other subjects:

Mathematics, 16.09.2020 19:01

English, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Geography, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

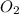 in the gas mixture the diver breathes in order to have the same partial pressure of oxygen in his lungs as he would at sea level? Note that the mole fraction of oxygen at sea level is 0.209.
in the gas mixture the diver breathes in order to have the same partial pressure of oxygen in his lungs as he would at sea level? Note that the mole fraction of oxygen at sea level is 0.209.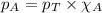 ........(1)
........(1)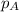 = partial pressure of oxygen at sea level = ?
= partial pressure of oxygen at sea level = ?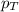 = total pressure at sea level = 1.00 atm
= total pressure at sea level = 1.00 atm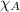 = mole fraction of oxygen at sea level = 0.209
= mole fraction of oxygen at sea level = 0.209