
Chemistry, 30.08.2019 07:30 seasmarie75
All of the following conditions of stp are true except a. 101.3 kpa b.3.81kpa. l/mol. k c. 24.2 l d. 273.15 k

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:50, shonnybenskin8
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1



Chemistry, 22.06.2019 17:10, sophiaa23
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1
You know the right answer?
All of the following conditions of stp are true except a. 101.3 kpa b.3.81kpa. l/mol. k c. 24.2 l d....
Questions in other subjects:




Social Studies, 09.03.2021 23:50

Mathematics, 09.03.2021 23:50

English, 09.03.2021 23:50

History, 09.03.2021 23:50


Biology, 09.03.2021 23:50

Mathematics, 09.03.2021 23:50

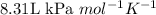 ,
, 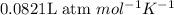 etc..
etc..

