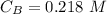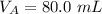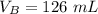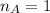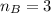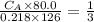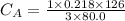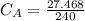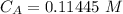Chemistry, 12.02.2020 20:06 liloleliahx2
The titration of 80.0 mL of an unknown concentration H3PO4 solution requires 126 mL of 0.218 M KOH solution. What is the concentration of the H3PO4 solution (in M)?

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 20:00, AaronEarlMerringer
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1

Chemistry, 22.06.2019 22:00, jespinozagarcia805
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a. rectant b. product c. supernate
Answers: 3
You know the right answer?
The titration of 80.0 mL of an unknown concentration H3PO4 solution requires 126 mL of 0.218 M KOH s...
Questions in other subjects:



English, 14.09.2021 06:00


History, 14.09.2021 06:00

Mathematics, 14.09.2021 06:00

History, 14.09.2021 06:00

Chemistry, 14.09.2021 06:00


History, 14.09.2021 06:00

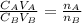
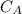 is the concentration of acid
is the concentration of acid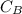 is the concentration of base
is the concentration of base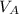 is the volume of acid
is the volume of acid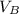 is the volume of base
is the volume of base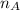 is the mole ratio of acid
is the mole ratio of acid 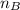 is the mole ratio of base
is the mole ratio of base