
Chemistry, 12.02.2020 05:28 friendsalwaysbae
For a ternary solution at constant T and P, the composition dependence of molar property M is given by: M = x1M1 + x2M2 + x3M3 + x1 x2 x3C where M1, M2, and M3 are the values of M for pure species 1, 2, and 3, and C is a parameter independent of composition. Determine expressions for M¯1,M¯2, and M¯3 by application of Eq. (10.7). As a partial check on your results, verify that they satisfy the summability relation, Eq. (10.11). For this correlating equation, what are the M¯i at infinite dilution?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:00, officialgraciela67
What is the volume occupied by 10.0 dm3 of gas at standard pressure after it has been compressedat constant temputure to 500.0 kpa?
Answers: 1

Chemistry, 22.06.2019 19:10, aamu15
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3
You know the right answer?
For a ternary solution at constant T and P, the composition dependence of molar property M is given...
Questions in other subjects:


Mathematics, 27.09.2020 16:01


Mathematics, 27.09.2020 16:01

English, 27.09.2020 16:01


Mathematics, 27.09.2020 16:01



History, 27.09.2020 16:01

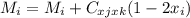 ...1
...1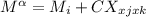 ...2
...2![M_{i} = [\frac{d(nM)}{dn_{i} }]_{P,t,n,j}](/tpl/images/0508/2556/4b4ae.png)


