

Answers: 2


Other questions on the subject: Chemistry



You know the right answer?
At a concentration of 1 M, the weak acid HNO 2 is 2% ionized, and the pH of the solution is 1.7. Wha...
Questions in other subjects:

English, 08.12.2021 01:00

Mathematics, 08.12.2021 01:00

Mathematics, 08.12.2021 01:00


English, 08.12.2021 01:00

English, 08.12.2021 01:00



Mathematics, 08.12.2021 01:00

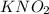 dissolved into the solution then the equilibrium will shift in the left direction.
dissolved into the solution then the equilibrium will shift in the left direction.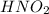 is:
is: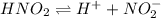
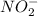 then the equilibrium will shift in the left direction.
then the equilibrium will shift in the left direction.

