Consider the reaction
H2(g) + 1 2 O2(g) → H2O(ℓ) + 286 kJ
How much H2 would...

Chemistry, 12.02.2020 04:42 gabrielbergemancat
Consider the reaction
H2(g) + 1 2 O2(g) → H2O(ℓ) + 286 kJ
How much H2 would have to be burned to yield America’s daily energy share of 260,000 kcal? (1 cal = 4.184 J)
1.) 1900 mol
2.) 3800 mol
3.) 7600 mol
4.) 1 mol

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, anthony4034
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 06:00, nikejose11
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3

Chemistry, 22.06.2019 06:30, backup5485
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 08:30, microwave13016
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1
You know the right answer?
Questions in other subjects:

English, 13.10.2019 21:10

Chemistry, 13.10.2019 21:10

Health, 13.10.2019 21:10

History, 13.10.2019 21:10


English, 13.10.2019 21:10

English, 13.10.2019 21:10

English, 13.10.2019 21:10

Computers and Technology, 13.10.2019 21:10

Mathematics, 13.10.2019 21:10

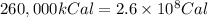 (Conversion factor: 1 kCal = 1000 Cal)
(Conversion factor: 1 kCal = 1000 Cal)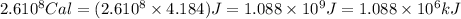
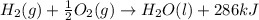
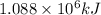 of energy will be released when
of energy will be released when 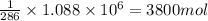 of hydrogen gas is consumed
of hydrogen gas is consumed

