Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, 2024daisjavien
Which actions would increase the rate at salt dissolves in water? stir the water? crush the salt? use less water? heat the water? cool the salt
Answers: 3

Chemistry, 22.06.2019 21:30, starl0rd211
Describe at least two advantages and two disadvantages of using hydropower as a source of energy.
Answers: 2

Chemistry, 22.06.2019 22:00, aliciaa101
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1
You know the right answer?
A sample of octane burns releasing 2290 J of heat to the surroundings, and the gases produced expand...
Questions in other subjects:




History, 24.01.2021 09:10

Mathematics, 24.01.2021 09:10





English, 24.01.2021 09:20

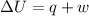
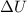 = internal energy
= internal energy